NUAGE
FILLER
Type 01
 Material Injected into skin or dermins to Improve or correct wrinkles, dents, acne scars or to contour facial line.
Material Injected into skin or dermins to Improve or correct wrinkles, dents, acne scars or to contour facial line.
FILLER
Type 01
 Material Injected into skin or dermins to Improve or correct wrinkles, dents, acne scars or to contour facial line.
Material Injected into skin or dermins to Improve or correct wrinkles, dents, acne scars or to contour facial line.


· Certified item
· Tissue Repair Biomaterial(B04230.01[4])
· Products Name : NUAGE
· Certification Number No. 6091
· Storage : Store at 2ºC~25ºC temperature, avoid light,
do not freeze
· Indication : Temporarily correct facial wrinkles by injecting Lidocaine containing cross-linked HA into dermis.
(Inspection Organization : Korea Testing & research institute)
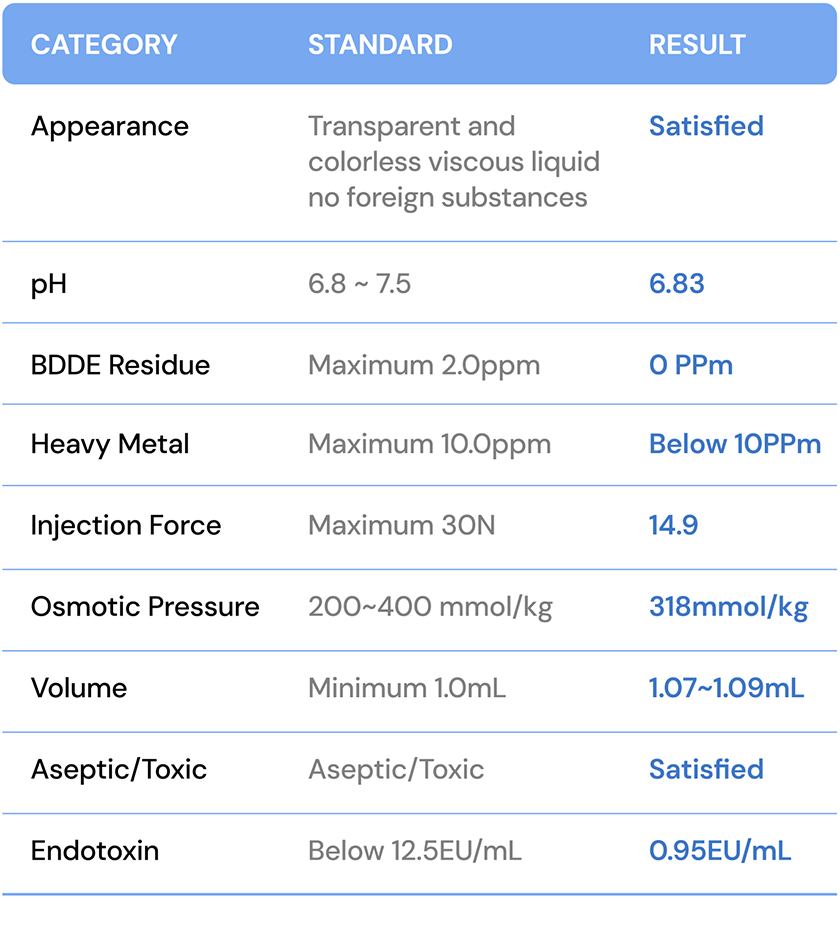
| CATEGORY | STANDARD | RESULT |
|---|---|---|
| Appearance | Transparent and colorless viscous liquid with no foreign substances | Satisfied |
| pH | 6.8 ~ 7.5 | 6.83 |
| BDDE Residue | Maximum 2.0ppm | 0 PPm |
| Heavy Metal | Maximum 10.0ppm | Below 10PPm |
| Injection Force | Maximum 30N | 14.9 |
| Osmotic Pressure | 200~400 mmol/kg | 318mmol/kg |
| Volume | Minimum 1.0mL | 1.07~1.09mL |
| Aseptic/Toxic | Aseptic/Toxic | Satisfied |
| Endotoxin | Below 12.5EU/mL | 0.95EU/mL |
Ideal Monophasic product manufactured by using special grinding machine,
Consistent injection (Product has uniformity), Soft, uniform gel type, easier to mold to achieve natural result.

Biphasic

Monophasic (Nuage)
| BIPHASIC | VS | MONOPHASIC |
|---|---|---|
| Particle | Particle | Gel |
| · Good volumizing · Easy to maintain its form · Lower risk of overcorrection |
Advantage | · Spread smoothly · Easily infected |
| Inconsistent | Disadvantage | Low volumizing effect |
| Nose, Chin | Rcommended area | Forehead |

NUAGE is
Supreme & Pure monophasic cross linked HA soft tissue filler.
Performs by adding volume rto facial tissue, thus correcting wrinkles & folds,
restoring a smooth appearance to the face, and redefining contour of the face.
Keeps your skin moisturized, stimulates the regeneration of the aged cells, and
supports collagen & elastin to maintain healthy skin.
| Type | Aqua | Optima | Delicate | Intense |
|---|---|---|---|---|
| Composition | 24mg/ml HA, Lidocaine 0.3% | 24ml/ml HA, Lidocaine 0.3% | 24mg/ml HA, Lidocaine 0.3% | 24mg/ml HA, Lidocaine 0.3% |
| Purpose | Moisturizing, Pore Tightening |
Fine wrinkle correction |
Deep wrinkle Correction, Volumizing |
Volumizing, Contouring |
| Indication | Full face | Periorbital Line, Lip contour |
Glabella Lines, Nasolabial folds, Marionette lines, Nose/forehead contour |
Nasolabial folds, chin, body contour |
| Injection Depth | Superficial Dermis | Mid Dermis | Mid, Deep Dermis | Subcutaneous |
| Duration | 3 month | 3~6 month | 6~12 month | Over 12 month |
| Package Unit | 1.1 ml | |||
| Needle Size | 30G (UTW) | 30G (UTW) | 27G (UTW) | 25G (UTW) |
| Storage | 2~25℃ | |||
| Complex Viscosity | Below 12.5EU/mL | 0.95EU/mL | 0.95EU/mL | 0.95EU/mL |
| BDDE | Not detected | |||
| Endotoxin | < 0.10 EU/ml (Acceptance Criteria <12.5 EU/ml) | |||
* BTW (Ultra Thin Wall needle)
: The Ultra Thin wall needle has a larger inner diameter compared to regular needles. It improves flow rates and lowers extrusion force during injection.
FILLER
Type 02
